Tetpor Plus filter cartridges have been specifically designed for the filtration of liquids and gases in the bulk pharmaceutical, chemical and biopharmaceutical industry where particulate removal, bioburden reduction and guaranteed sterility is required.
The increasing use of ozonation for the treatment of WFI systems has highlighted compatibility issues with vent filters based on standard polypropylene components. The introduction of a fully validated 0.2-micron sterilizing grade TETPOR PLUS filter cartridge provides guaranteed long term performance in these applications with the additional benefit that the water intrusion test method can validate the filters integrity.
The high voids volume single layer PTFE membrane ensures an excellent combination of flow rate and retention.
Features and Benefits- Sterile filtration of oxygen / oxygen enriched feeds in cell culture
- Exceptional resistance to solvents and oxidative environments
- Ideal for sterile venting on ozonated water systems
- Fully validated to ASTM F838-83 for sterilizing grade filters
- PTFE membrane
- Available in a wide range of micron ratings to suit all applications
SPECIFICATIONSMaterials of Construction- Filtration Membrane: Polytetrafluoroethylene
- Upstream Support: Polytetrafluoroethylene
- Downstream Support: Polytetrafluoroethylene
- Inner Support Core: PFA
- Outer Protection Cage: PFA
- End Caps: PFA
- Standard o-rings: FEP Encapsulated, Silicone
Food and Biological SafetyMaterials conform to the relevant requirements of 21CFR Part 177, EC1935 / 2004 and current USP Plastics Class VI - 121 °C and ISO10993 equivalents.
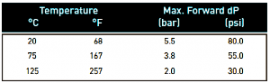
Recommended Operating ConditionsUp to 125 °C (257 °F) continuous operating temperature and higher short-term temperatures during CIP to the limits on the table to the left.
Effective Filtration Area (EFA)10¨ (250 mm) 0.63 m2 (6.78 ft2)
K Size (125 mm) 0.32 m2 (3.44 ft2)
Cleaning and SterilizationTetpor Plus cartridges can be repeatedly steam sterilized in situ or autoclaved at up to 142 °C (287.6 °F) for a maximum of 30 cycles. For detailed operational procedures and advice on cleaning and sterilization, please contact Technical Support.
Retention CharacteristicsTetpor Plus filter cartridges are validated by bacterial challenge testing with Brevundimonas diminuta to current ASTM F838-05 methodology (107 organisms / cm2 EFA minimum) with typical in-house
challenge levels being 1011 organisms per 10¨ (250 mm) module.
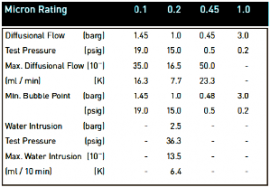
Integrity Test DataThe following is the integrity test information for the micron ratings available within the Tetpor Plus product range. Diffusional flow and bubble point values are given for cartridges wetted in 60:40 v/v IPA:Water solution.
Pharmaceutical ValidationA full validation guide is available upon request from Laboratory Services Group (LSG).
Download the Parker Domnick Hunter Tetpor Plus Filter Cartridges Datasheet Talk to one of our filtration experts today at
(855) 737-4717, or fill out our
online form to learn more.